Room temperature CO2 reduction catalyst
2019-04-05 11:18 by Ian
I've seen lots of media outlets in Normistan touting a new liquid phase catalyst as a means of removing carbon dioxide from the atmosphere and sequestering it as coal. But my chemistry background tells me that this is never going to be useful for that purpose.
What follows is my rough analysis of the energy requirements of running this reaction with the given catalyst.
Here is the paper that that the journalists are reporting about.
Assumptions
I didn't bother adjusting my calculations for the less-than-unity faradic efficiency of the electrochemical cell. This has the effect of making my energy estimates a best-possible case. No real-world cell will ever be as good as my calculations suggest.
I also assume that an average human produces 2.3 lbs of CO2 per day. Please forgive the non-scientific source, but as you will see, it won't change the order-of-magnitude of the results.
The claim coming from Normistan
By using liquid metals as a catalyst, we've shown it's possible to turn the gas back into carbon at room temperature, in a process that's efficient and scalable.
While more research needs to be done, it's a crucial first step to delivering solid storage of carbon.
The nature of the electrochemical cell
It's cerium oxide dissolved in a gallium alloy, with dimethylformamide as the CO2 solvent. There also appears to be (at least) tin in the electrode alloy, for the sake of lowering the melting point of the electrode.
They think that the conversion is happening in two steps. They first have to "warm up" the electrode to reduce the oxygen off the cerium (which, like almost all metals "rusts" spontaneously in the presence of oxygen). After that, it looks like the oxygen from the CO2 is transferred to the reduced cerium, which then needs to be reduced again.
So.... they need 4 mol of electrons to reduce 1 mol of carbon. It appears that their reference electrode was silver, which is why they are giving the half-reaction potential in terms of Ag/Ag+. Each successive reduction on cerium will cost progressively more, but they appear to have averaged it out to 1.2v above Ag/Ag+ (0.8v). Thus, they applied ~2v to the system.
Now, for the thermodynamics....
CO2 weighs 44.01 g/mol. 12.01g of that is carbon.
To reduce that much CO2 will cost....
Which simplifies to....
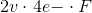
...where F is the Faraday constant (which was redefined this year, apparently).
This comes out to about 771.882 kilojoules. Since 1 joule is 1W/sec, we can put that amount of energy into perspective with a car motor metaphor. This is like running a 1034.69 horsepower motor for 1 second.
Put less circuitously, you need to apply 1034.69 horsepower to that electrochemical cell to remove 44.01g of CO2 from the solvent every second, and producing 12.01g of solid carbon and 36g of oxygen as products.
It might be useful to (pull CO2 out of) and (replenish the O2 in) a confined space like a submarine or a manned spacecraft, thus saving consumables. But again: Only if electricity were so cheap that you were already throwing it away. Let's see if it's even useful in that context.
Apparently, the average human produces 1044.2g of CO2 every day. Dividing by the molar mass of CO2, we get about 23.73 moles of CO2.
That's about 18.314 megajoules per day required to make the average human carbon neutral. This easily rules out its use on spacecraft. And for their crew sizes, even nuclear submarines. Dividing over time to arrive at a wattage, I find 212 watts.
So there you have it. If you can use your body to power a hand-crank that generates at least 212 watts, you can make yourself carbon neutral, and that assumes you never sleep, and can do it without breaking a sweat.
Put another way, the Palo Verde nuclear power plant is capable of generating 3,937 megawatts of electricity. If this entire plant's output was devoted to the task of offsetting the carbon output of humans, it would only account for 18,570,754 people.
If you wanted to use this catalyst to offset the carbon output of all of the 7.5 billion people on Earth, you would need to build 404 new nuclear power plants the size of Palo Verde.
Conclusion
It's an interesting catalyst because it trades reaction temperature for electrochemical potential. But it won't be used to remove CO2 from the atmosphere. Electricity is higher grade energy than heat, Thermodynamics won't be cheated, and there is no way to produce that much electricity at so little a carbon cost (unless you use nuclear).
The author's claim that the catalyst can be used to produce ultra-thin carbon sheets for industrial or scientific purposes is sound. It looks to me like it will in fact do this (at enormous energy cost).
If you want to remove CO2 from the atmosphere, plant a tree.
Journalists and politicians generally have no grasp of economics or science. Anyone telling you that this is going to be used to "battle climate change", or sequester carbon en masse doesn't know enough to have a valid opinion about it. My advice would be to change your media outlet.
Previous: Professional advice for new engineers
Next: Recent breakout boards